|
Polymer
Structure
Although the fundamental property of bulk
polymers is the degree of polymerization, the physical structure
of the chain is also an important factor that determines the
macroscopic properties.
The terms configuration and
conformation are used to describe the geometric structure of
a polymer and are often confused. Configuration refers
to the order that is determined by chemical bonds. The
configuration of a polymer cannot be altered unless chemical
bonds are broken and reformed. Conformation refers to
order that arises from the rotation of molecules about the single
bonds. These two structures are studied below.
Configuration
The two types of polymer configurations are
cis and trans. These structures can not be changed by
physical means (e.g. rotation). The cis configuration
arises when substituent groups are on the same side of a
carbon-carbon double bond. Trans refers to the
substituents on opposite sides of the double bond.
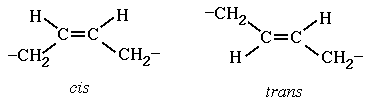
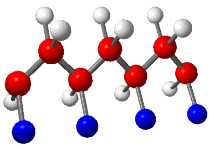
Stereoregularity is the term used to describe
the configuration of polymer chains. Three distinct structures
can be obtained. Isotactic is an arrangement where all
substituents are on the same side of the polymer chain. A
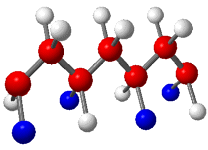
syndiotactic polymer chain is composed of alternating
groups and atactic is a random combination of the
groups. The following diagram shows two of the three
stereoisomers of polymer chain.
|
 |
|
 |
|
Isotactic |
|
Syndiotactic |
Conformation
If two atoms are joined by a single bond then
rotation about that bond is possible since, unlike a double bond,
it does not require breaking the bond.
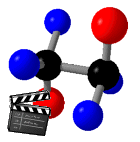
The ability of an atom to rotate this way
relative to the atoms which it joins is known as an adjustment of
the torsional angle. If the two atoms have other atoms
or groups attached to them then configurations which vary in
torsional angle are known as conformations. Since
different conformations represent varying distances between the
atoms or groups rotating about the bond, and these distances
determine the amount and type of interaction between adjacent
atoms or groups, different conformation may represent different
potential energies of the molecule. There several possible
generalized conformations: Anti (Trans), Eclipsed (Cis), and
Gauche (+ or -). The following animation illustrates the
differences between them.
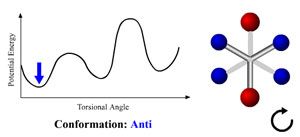
Conformation
Lattice Simulation
Like the polymer growth simulation, the
conformation lattice simulation takes a statistical approach to
the study of polymers. Probabilities of the different
conformations are assigned which produces a polymer chain with
many possible shapes. Click the icon to enter the virtual
laboratory.
Other Chain
Structures
The geometric arrangement of the bonds is not
the only way the structure of a polymer can vary. A branched
polymer is formed when there are "side chains" attached to
a main chain. A simple example of a branched polymer is shown in
the following diagram.
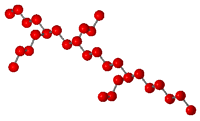
There are, however, many ways a branched
polymer can be arranged. One of these types is called "star-branching".
Star branching results when a polymerization starts with a single
monomer and has branches radially outward from this point.
Polymers with a high degree of branching are called
dendrimers Often in these molecules, branches themselves
have branches. This tends to give the molecule an overall
spherical shape in three dimensions.
A separate kind of chain structure arises when
more that one type of monomer is involved in the synthesis
reaction. These polymers that incorporate more than one kind of
monomer into their chain are called copolymers. There
are three important types of copolymers. A random copolymer
contains a random arrangement of the multiple monomers. A
block copolymer contains blocks of monomers of the same
type. Finally, a graft copolymer contains a main chain
polymer consisting of one type of monomer with branches made up
of other monomers. The following diagram displays the different
types of copolymers.
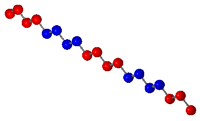 |
Block Copolymer |
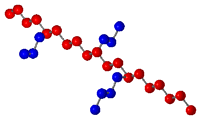 |
Graft Copolymer |
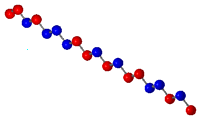 |
Random Copolymer |
An example of a common copolymer is Nylon.
Nylon is an alternating copolymer with 2 monomers, a 6 carbon
diacid and a 6 carbon diamine. The following picture shows one
monomer of the diacid combined with one monomer of the diamine:
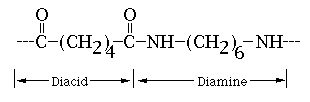
Cross-Linking
In addition to the bonds which hold monomers
together in a polymer chain, many polymers form bonds between
neighboring chains. These bonds can be formed directly between
the neighboring chains, or two chains may bond to a third common
molecule. Though not as strong or rigid as the bonds within the
chain, these cross-links have an important effect on
the polymer. Polymers with a high enough degree of cross-linking
have "memory." When the polymer is stretched, the cross-links
prevent the individual chains from sliding past each other. The
chains may straighten out, but once the stress is removed they
return to their original position and the object returns to its
original shape.
One example of cross-linking is
vulcanization . In vulcanization, a series of cross-links
are introduced into an elastomer to give it strength.
This technique is commonly used to strengthen rubber.
Classes of
Polymers
Polymer science is a broad field that includes
many types of materials which incorporate long chain structure of
many repeat units as discussed above. The two major polymer
classes are described here.
Elastomers,or rubbery materials,
have a loose cross-linked structure. This type of chain structure
causes elastomers to possess memory. Typically, about 1 in 100
molecules are cross-linked on average. When the average number of
cross-links rises to about 1 in 30 the material becomes more
rigid and brittle. Natural and synthetic rubbers are both common
examples of elastomers. Plastics are polymers which,
under appropriate conditions of temperature and pressure, can be
molded or shaped (such as blowing to form a film). In contrast to
elastomers, plastics have a greater stiffness and lack reversible
elasticity. All plastics are polymers but not all polymers are
plastics. Cellulose is an example of a polymeric material which
must be substantially modified before processing with the usual
methods used for plastics. Some plastics, such as nylon and
cellulose acetate, are formed into fibers (which are regarded by
some as a separate class of polymers in spite of a considerable
overlap with plastics). As we shall see in the section on liquid
crystals, some of the main chain polymer liquid crystals also are
the constituents of important fibers. Every day plastics such as
polyethylene and poly(vinyl chloride) have replaced traditional
materials like paper and copper for a wide variety of
applications. The section on Polymer Applications will go into
greater detail about the special properties of the many types of
polymers.
Thermal Properties of Polymers
More 8
| 

 Plastics Engineering
Plastics Engineering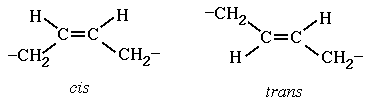








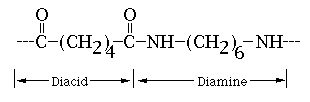



 Knowledge Base
Knowledge Base

















 Knowledge Base
Knowledge Base


